Another Round of Bullshit Bingo

If like me, you call “bullshit” whenever a news article claims that “e-cigs are as bad as smoking”, you’ve probably been playing the same game of bullshit bingo as I have.
Trouble is, I never seem to win anything.
The latest headlines, generated by a dodgy press release naturally, to spread like wildfire is, of course, all about “toxic metals” found in the liquid and aerosol. Naturally, when I first saw the headline, I uttered the now infamous phrase: “I call bullshit”.
Here’s why.
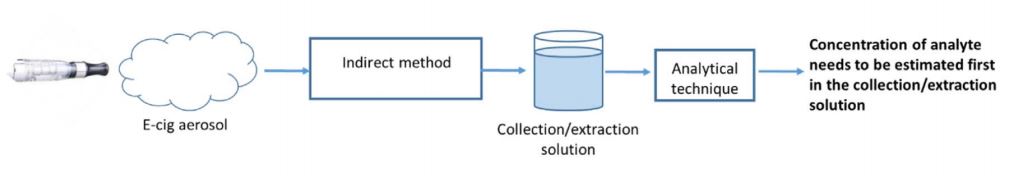
Let’s start with the collection of the aerosol itself. In most cases, the aerosol is collected via an indirect method:
Olmedo P, Navas-Acien A, Hess C, Jarmul S, Rule A. 2016. A direct method for e-cigarette aerosol sample collection. Environ Res 149:151–156
That is, the aerosol is filtered or directly diluted into a collection solution; the baseline constituents are known prior to analysis and then the solution is analysed for detectable levels of a particular analyte.
This type of collection and analysis is routine and widely recognised as the most accurate.
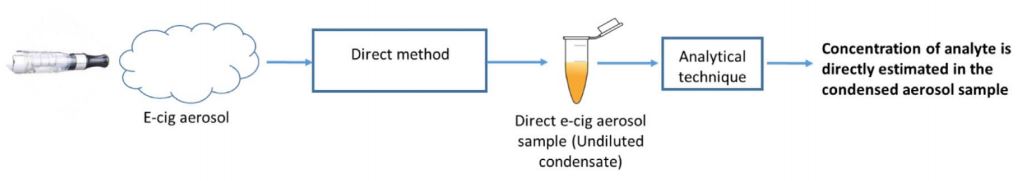
The method used in this study doesn’t filter the aerosol, nor is there a known baseline solution used in the analysis. Instead, the aerosol is condensed immediately after generation into a container (pipette tips are generally used, though it is likely alternative containers are also used). This is known as the direct method.
Olmedo P, Navas-Acien A, Hess C, Jarmul S, Rule A. 2016. A direct method for e-cigarette aerosol sample collection. Environ Res 149:151–156
The immediate problem here is, of course, the collection container. Without a baseline sample, there’s no way to determine if the sample collected has been contaminated.
The second problem with the aerosol collection, is, of course, the puff topography.
Briefly, a peristaltic pump placed inside a fume hood puffed the e-cigarette and the generated aerosol was collected in a 1:5-mL centrifuge tube via deposition in a series of conical pipette tips and plastic tubing (1 L=min, 4 s per puff and 30-s interpuff time).
As one astute Redditor pointed out:
The average human breath is 500 ml and takes place over 1.5-2 seconds. The flowrate that humans breathe in, on average, is then 15-20 L/min, and 15-20 times faster than the test concluded.
This, of course, highlights the most obvious flaw, the dry puff.
The simple fact is, at the aerosol generation at 1L per minute is far too slow, bringing airflow into the equation. As I pointed out on the Juice Monster study, airflow plays a critical role in vaping - alongside the other obvious factors such as wattage, coil resistance and coil construction.
Once again, tobacco control researchers; or specifically researchers funded by tobacco control outlets, have performed a study that ignores the central feature of vaping - the user.
Further flaws in this study, as highlighted by the redoubtable Carl Phillips and Tom Pruen include how these researchers came up with the various concentrations.
But I’ve almost forgotten the best bit.
In tank samples we found statistically significant differences by voltage tertiles for Al, Fe, and Mn, with the intermediate tertile presenting the highest metal concentrations.
As almost everyone who follows this debate should know, when you apply current to a resistant material, it gets hot. When that material gets too hot, it can start to degrade - thermal degradation. When this happens, pieces of that resistant material breakdown, and if said material is surrounded by a liquid then naturally pieces of the resistant material will end up in the liquid.
It isn’t rocket science, yet these researchers are acting like they’ve discovered the Holy Grail. As we know from previous work by Dr Farsalinos, the formation of aldehydes occurs under intense heat situations - the “dry puff”. Most of these intense heat situations occur at 4V and higher.
Now, if you’ve been paying attention, I’ll be willing to bet you can guess what voltages these genius researchers tested at, can’t you?
Yep. 4V to 4.4V.
For Ni, the difference by voltage was borderline significant (p= 0:05) with concentrations also higher at the intermediate tertile (4.00–4.40 V).
These are just some of the flaws I spotted in the main text. What flaws did the researchers mention in their discussion?
We also assumed that we collected the total weight of the emitted aerosol, although we know that around 20% remains in the tubing and around 10% of the aerosol is lost through the venting groove of the collection device.
So their figures are likely to be inaccurate or overestimated.
A reason for why our values are at least an order of magnitude higher is that mainstream aerosol has not undergone mixing in indoor air like secondhand aerosol.
Fools! Indoor air is drawn through the tank and over the coil during inhalation. With the regular human breath, this occurs far faster than in your scenario.
In our study, metal concentrations tended to be lower in aerosol than in tank samples. Correlations between concentrations of different metals were lower in the aerosol than in the tank. We do not have a definite explanation for these differences, but metal concentrations in the tank e-liquid cannot be expected to be equal to those in the aerosol for the following reasons: a) Mass transfer of metal compounds into the aerosol can be expected to be metal specific. b) Some of the metals have been shown to exist as solid beads within the aerosol droplets, and it is hypothesized that the beads originate from metallic e-cigarette components such as the heating coil.
Here’s something the media frenzy missed completely. The concentrations, low as they are, in the aerosol are significantly lower in the aerosol than in the tank itself. Well, duh. Of course, they would be.
Y’see, there’s another element to an e-cig that is often overlooked by researchers - the wick. Many of the metal particulates - although that’s not strictly the right term here - will get caught on the wick as the coil is often (though not always) surrounded on all “sides” by it.
At the beginning of our collection process, (within the first puffs), when drops are starting to be formed inside the tubing, more droplets in the 300–500 nm range will escape from the collection device than larger and smaller droplets, which are more efficiently collected on the device walls due to the processes of impaction and diffusion, respectively.
Another “well, duh” moment. At the start of any vaping session, the liquid and coil will be ‘cold’, therefore - as with any heat-related device - the initial phase is highly inefficient, and in the case of vaping, the liquid doesn’t vapourise as quickly.
Unlike many other studies, this one doesn’t report which devices were actually used, other than a vague mention of “58 participants to bring their regular e-cigarette device and refilling dispenser”. As these participants were recruited from vape conventions and vape shops, it is likely that these participants were using more ‘advanced’ devices, rather than the researchers favourite - the CE4.
As Dr Farsalinos states:
The “significant amount” of metals the authors reported they found were measured in μg/kg. In fact they are so low that for some cases (chromium and lead) I calculated that you need to vape more than 100 ml per day in order to exceed the FDA limits for daily intake from inhalational medications. The authors once again confuse themselves and everyone else by using environmental safety limits related to exposure with every single breath, and apply them to vaping. However, humans take more than 17,000 (thousand) breaths per day but only 400-600 puffs per day from an e-cigarette.
Many, many flaws with a fixation on comparing the levels detected with health/regulatory levels and no control for mass loss from the coils. All to suit the current US regulatory landscape.
Remember, we’re on the side of the angels here. This lot, well, I’d call them “muppets” but that’s being unkind to actual muppets.