Snus and the EU

The European Union doesn’t have much love for tobacco at all, in fact it does everything it can to make life as miserable as possible for those they think are daft enough to smoke. Not content with pushing up prices on all forms of tobacco in a vain effort to get people to quit, the EU has undertaken a massive regulatory campaign to make it impossible or next to impossible for reduced risk products to be made available. Electronic cigarettes are only the latest in the crusade against all forms of “tobacco” use; despite of course the fact that e-cigarettes don’t actually contain tobacco, but they do mimic the act of smoking (part of the reason for the success for many users).
Smokeless tobacco products have had a lot more problems. ;Since 1992, Snus has been banned throughout the Union - three years before Sweden joined in 1995 - with the main reason (natch) being “a fear that it could become a ‘gateway’ to smoking for young people” - where have we heard that before?
The EU snus ban, under directive 92/41/EEC - which was supposedly concerning the labelling of tobacco products, specifically Article 8a, prohibited any tobacco for oral use to be ;placed on the market. This ban was then reaffirmed in 2001, again ;under Article 8 it was abundantly clear:
Member States shall prohibit the placing on the market of tobacco for oral use, without prejudice to Article 151 of the Act of Accession of Austria, Finland and Sweden.
That position hasn’t changed, despite overwhelming evidence to support Snus as a viable, substantially less harmful alternative to smoking traditional combustible tobacco. Yet the EU, in its “infinite wisdom” decided to renew the bloc wide ban again under the revised Tobacco Products Directive in 2014.
Swedish Match, one the leading manufacturers of Snus, has tried - particularly in the US - to have Snus recognised as a valid ;harm reduction product:
In 2014, Swedish Match sought to:
- Remove the warning, “This product can cause gum disease and tooth loss”;
- Remove the warning, “This product can cause mouth cancer”; and,
- Replace the warning, “This product is not a safe alternative to cigarettes” with this text: “No tobacco product is safe, but this product presents substantially lower risks to health than cigarettes.”
As you will no doubt guess, the FDA rejected the application as covered in great detail by Brad Rodu here, despite decades of research on Snus coupled with the simple fact that in Sweden (where Snus is most prevalent) the smoking prevalence is the lowest - as of 2015 it was 11.7% - along with the lowest level of tobacco-related mortality and the highest rate of smokeless tobacco use. Coincidence?
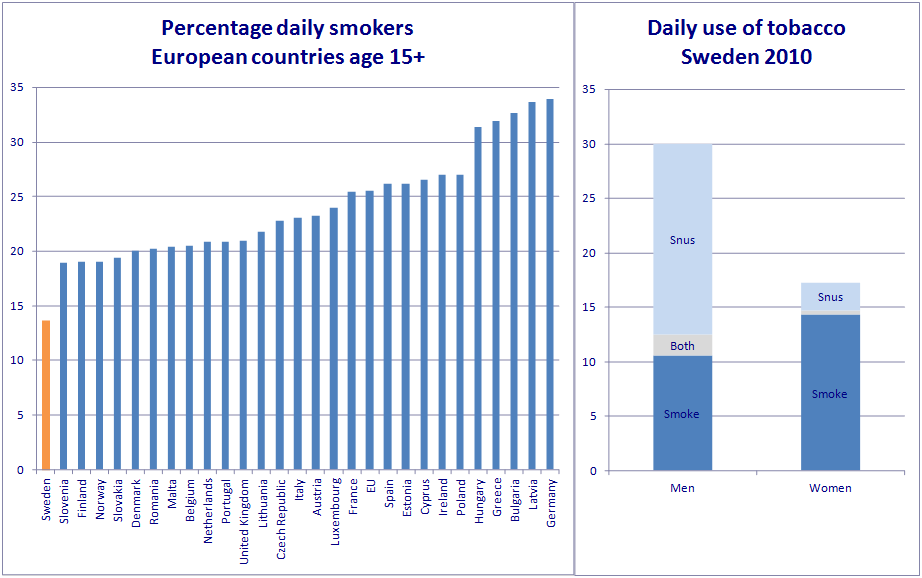
Graphs courtesy of Clive Bates
However, the fight to have Snus recognised as a viable harm reduction product - which many in the US and EU utterly fail to recognise it as - is far from over. July 2016 Swedish Match announced a legal action to overturn the EU wide Snus ban. The TPD, while being harsh on e-cigarettes, is worse than draconian for snus. There is sufficient evidence, more than sufficient in actual fact, to have the ban on sale of snus overturned.
Swedish Match followed the Pre-Market Tobacco Application (PMTA) to allow for a new range of snus to be placed on the US market to bring the products sold in the US from their 2011 versions to products that are the same as (or at least more similar to) the products being sold in Sweden. While the FDA did not authorise a reduced risk claim on the products it did, however, mean that the product had been evaluated as “appropriate for the protection of public health”, and seeing as a large proportion of the evidence base submitted with the PMTA came from Europe it does make the EU-wide snus ban all the more idiotic.
Of course, Swedish Match have tried legal action before in 2003 - after the previous TPD came into force - where the ECJ decided that snus was a “new” tobacco product. That summary is blighted by the inclusion of notification of “novel tobacco products” in the revised TPD in 2014. As Clive points out:
The position is now that either:
- snus is discriminated against because, absurdly, it is treated differently to smokeless tobacco products that are chewed rather than sucked.
- or snus is discriminated against because it is treated differently to other novel tobacco products.
There is no escape from this – the court’s ‘novel product’ rationale from the 2003 case based is gone. Of course, snus isn’t a novel product – it is widely known and well-understood and should be treated as any other smokeless tobacco product.
The weight of evidence, not to mention authorisation from the FDA to allow “modified” products to be sold, is clearly in favour of Swedish Match to repeal the snus ban, at least in the UK. This then can set a precedent not just for snus, which does carry some risk but only a fraction compared with tobacco, but for e-cigarettes also. If a reduced risk product like snus can have its ban lifted in light of the substantial evidence (which amusingly even the World Health Organisation recognises), it marks a clear start in the recognition and benefits of tobacco harm reduction in a place where once there was none.
Furthermore, the New Nicotine Alliance ;have asked the court to be allowed to make its own submission in order to provide a consumer perspective on the ban and of the benefits that will follow if the ban is lifted.
Buckle up Dorothy, Kansas is going bye-bye!